Clinical Data Management
Meet Marvin EDC
As of today, more than 1,500 studies worldwide have been managed using Marvin EDC.
Marvin is one of the most comprehensive eClinical solutions on the market.
Marvin provides intuitive, easy to use tools that do not require programming knowledge. With Marvin clinical trials become easy, streamlined and cost-effective. We have custom-made solutions for CROs and for academic institutions, too.
Why choose Marvin to run your Trial?
Marvin provides solutions for pharmaceuticals, biotech, medical devices, cosmetics, and consumer health trials. It is easy to set up and users praise its usability and multiple integrated features.
FLEXIBLE FOR ANY TYPE OF STUDY
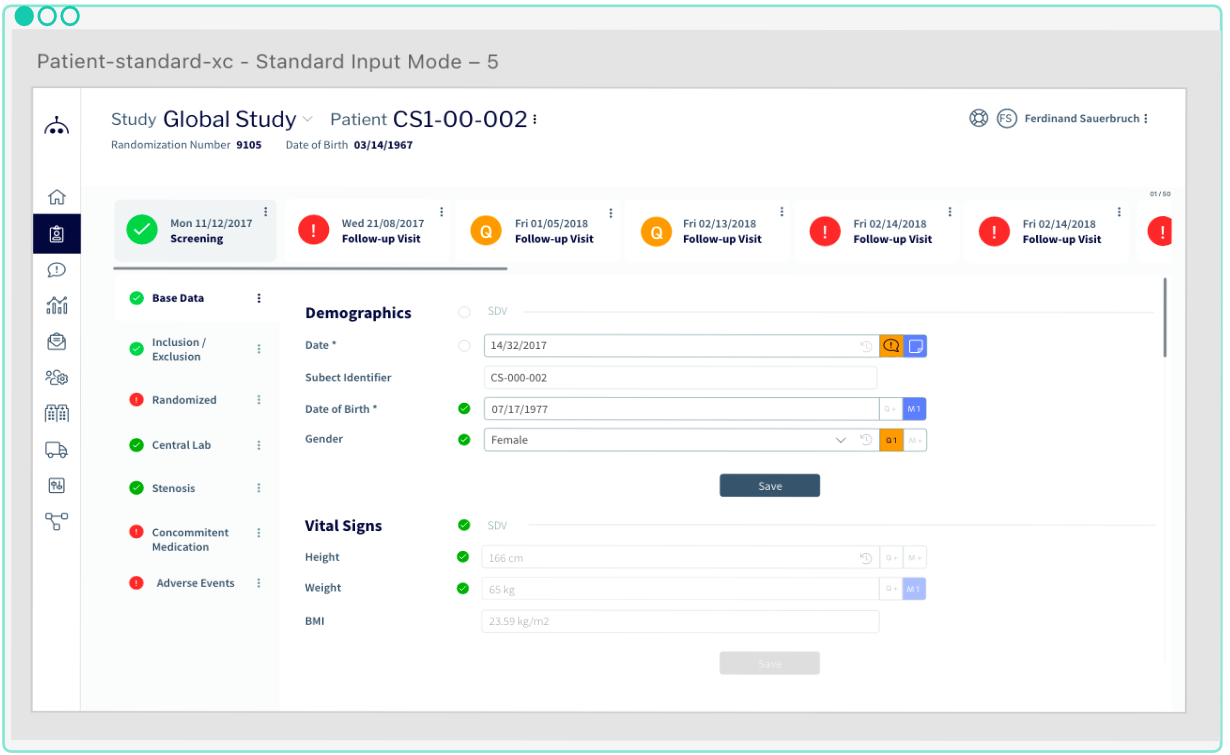
Easy Study Set-Up and Management
Enjoy a well-built and flexible modern application to safely collect your clinical data. A versatile software that enables easy study set-up and management. Both customers and end users value the simplicity combined with complex, customizable workflows. The integrated solution comprised of various eClinical modules, optimally supports clinical trial processes and enables seamless data flow.

StudyMetrIQs – for more insights in your study data
Get the clarity you need, from high-level KPIs to detailed operational metrics, right inside Marvin EDC.
Smarter Oversight. Better Decisions.
StudyMetrIQs empowers your team to track progress and make data-driven decisions effortlessly.
Built for Marvin 3.0
Seamlessly integrated with Marvin 3.0, StudyMetrIQs delivers customizable, interactive dashboards that bring your study data to life.
See More. Know More. Do More.
With StudyMetrIQs, gain real-time visibility into your clinical study data, all in one powerful dashboard solution.
Benefits
- Create unlimited dashboards in your layout
- Embed different reporting widgets in one dashboard
- Available within your Marvin EDC environment
- Do-it-yourself editor → Ad-hoc Reporting with no training requirement

69
Marvin is used in 69 Countries
worldwide
1500
More than 1200 studies realized
with Marvin EDC
BUILT FOR GLOBAL BUSINESS
Impressive universal Capabilities
Most Clinical Trials are conducted internationally spanning multiple countries or even continents. Marvin is designed with a comprehensive vision of the challenges global studies bring. Beginning with a centralized database that is accessible from any web browser, the addition of not just multi-language, but every language became a standard offering. Sensitive to the fact that not all regions have internet access, Marvin of course also allows for double-data entry (DDE) and hybrid studies.
Strong Interfacing
Marvin – Beyond Data Capture
- Quick and easy implementation of mid study changes
- Advanced query management
- Flexible for any type of study
- Complete Audit Trail
- (S)AE Notifications
- Electronic Signatures

All in One eClinical Software Platform
Scope of Features


Coder
Classify safety-related data such as adverse events or concomitant medications. Coder supports standard MedDRA and WHODrug dictionaries to automatically and manually encode verbatim terms by use of powerful search mechanisms. The coder module is fully integrated with Marvin via a Web Service API. Hence it is possible to utilize it in other solutions (or EDC systems). Anywhere, anytime: The coder can be accessed any time and place, meaning you can code at your convenience
- Customizable standard user roles and workflows
- Intuitive user interface
- Upgrade dictionary during study conduct
- Automated term matching
ePRO
Due to the increasing importance of Patient Reported Outcomes (PRO) in all types of studies we have a state-of-the art ePRO module in Marvin. Data will be captured in the same database as all eCRF forms. This facilitates real-time reporting including various wearable devices.
- Integrated as part of your clinical study
- Works on any device; no app needed
- Supports EQ5D questionnaires
- Easy to setup
- Utilize existing libraries from the composer
Randomization and Trial Supply Management
Our randomization system is an IWRS (Interactive Web Response System) integrated into Marvin and does not require an interface to any other external systems. It offers open or double-blind, multi-strata, multi-group randomization based on a block list or a widely configurable range of algorithms like variance minimization. We added CARA randomization to support multi-armed clinical trials. We are ready for adaptive trial design, so your trial is more efficient and ethical.
Adaptive designs can be applied across all phases of clinical research, from early-phase dose escalation to confirmatory trials.
- Seamless integration with our EDC
- Advanced materials management
- Offers multiple randomization methods

Expert Resources
Marvin is a universal software platform with multiple features to better manage the whole lifecycle of your clinical trial. “Quick set-up” is named top-3 benefit of Marvin compared with other EDC tools. Find out more.